Emerging forms of immunotherapy include Adoptive Cell Transfer (ACT) which utilise the patient's own immune cells to target the cancer. The most advanced method of these options when it comes to clinical development is CAR T-cell therapy. CAR T-cell therapy has produced positive results in some patients, both children and adults who have been unsuccessful with other forms of treatment.
What is CAR-T Cell Therapy?
CAR-T cell therapy is a new form of genetic or immunotherapy used to treat cancer by strengthening the patient's immune system to fight the cancer.
When fighting disease, the body creates T-cells which find and destroy the cells responsible for the disease.
In cases where the body is not able to combat the disease, this may be due to insufficient creation of T-cells, the inability of the immune system to create the required T-cell, or the inability of the T-cell to locate the cancer.
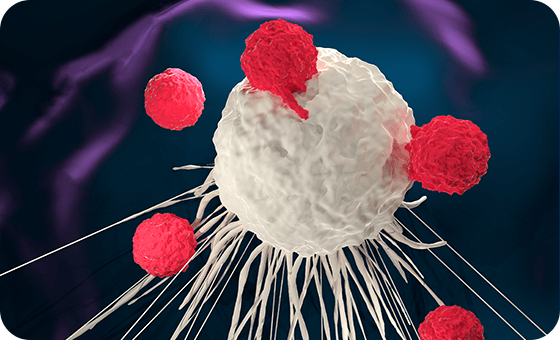
In CAR-T cell therapy, Chimeric Antigen Receptors (CAR) are engineered to infuse with the patient’s existing T-cells, programming them to recognise and bind to the cancer cells.
An activation domain engineered into the cell then functions to activate the T-cell to combat the cancer. Simply, the treatment involves enhancing existing cells to identify and fight the cancer.
What types of cancers is CAR-T Cell Therapy currently being developed for?
- Non-small cell lung cancer
- Colorectal cancer
- Prostate cancer
- Ovarian cancer
- Pancreatic cancer
- Gastric carcinoma
- Esophageal carcinoma
- Glioblastoma
- Hepatocellular cance
- Breast cancer
- Neuroblastoma
- Renal cell carcinoma
- Urothelial carcinoma
- Cervical carcinoma
- Mesothelioma
- Anaplastic thyroid carcinoma
- Melanomas
- Sarcomas
What does CAR-T Cell Therapy
involve for patients?
CAR-T cell therapy is a simpler technique to go through than many treatments. When it works well, CAR-T cell therapy may keep controlling a cancer or disease for years.
For most CAR-T cell therapies, blood collection will be the first clinical step – to prepare genetically-engineered cells that can attack the cancer and strengthen the immune system. This is like giving a blood donation, only quicker and with much less blood drawn.
The treatment is usually prepared within two weeks of blood collection. The patient may then go through several days of preliminary chemotherapy, not to kill the cancer, but to make room in their body for re-infusion of CAR-T cells. This chemotherapy will typically be in a hospital clinic, but patients are likely to tolerate it well.
The CAR-T cell therapy phase then begins. Genetically-engineered cells are re-infused into the patient via a vein.
The patient usually feels well during this procedure. However, in the week that follows, some patients may become ill, with flu-like or respiratory symptoms.
Some patients can become very unwell after CAR-T cell therapy, with side effects such as major lung damage or brain disturbance requiring intensive care. There is a small risk of dying at this stage – but with supportive care, patients usually recover quickly, and are well within weeks.
Once patients are well, they can leave the hospital, have tests to see how their cancer or disease is responding, and hopefully return home. During this time, other treatments may also be utilised to enhance the effect of CAR-T cell treatment.
For many patients, the whole process may be complete within two months and, if it is working well, no further therapy may be required at that time.
ConnectGene can assess
if CAR-T cell therapy is right for you

CAR T-cell therapies have been approved by the Food and Drug Administration (FDA) for the treatment of acute lymphoblastic leukemia (ALL) in children and advanced lymphomas in adults.
ConnectGene can assist in assessing your suitability for CAR-T cell therapy, beginning with the initial screening process, through to personalised treatment selection. From here, we organise your referral to a treatment centre that has the capability to deliver experimental CAR-T cell therapy on a clinical trial.
Speak to our team today to organise the first step of this process to see if CAR-T cell therapy may be suitable for your situation.


